Cancer Treatment Reviews 107 (2022) 102405
Available online 5 May 2022
0305-7372/© 2022 The Authors. Published by Elsevier Ltd. This is an open access article under the CC BY-NC-ND license (http://creativecommons.org/licenses/by-
nc-nd/4.0/).
Anti-tumour Treatment
The paradigm shift in treatment from Covid-19 to oncology with
mRNA vaccines
Jiao Wei, Ai-Min Hui
*
Shanghai Fosun Pharmaceutical Industrial Development, Co., Ltd., 1289 Yishan Road, Shanghai 200233, China
Fosun Pharma USA Inc, 91 Hartwell Avenue, Suite 305, Lexington, MA 02421, USA
ARTICLE INFO
Keywords:
mRNA
Cancer vaccine
Covid-19
Oncology
Optimization
ABSTRACT
mRNA vaccines have gained popularity over the last decade as a versatile tool for developing novel therapeutics.
The recent success of coronavirus disease (COVID-19) mRNA vaccine has unlocked the potential of mRNA
technology as a powerful therapeutic platform. In this review, we apprise the literature on the various types of
cancer vaccines, the novel platforms available for delivery of the vaccines, the recent progress in the RNA-based
therapies and the evolving role of mRNA vaccines for various cancer indications, along with a future strategy to
treat the patients. Literature reveals that despite multifaceted challenges in the development of mRNA vaccines,
the promising and durable efcacy of the RNA in pre-clinical and clinical studies deserves consideration. The
introduction of mRNA-transfected DC vaccine is an approach that has gained interest for cancer vaccine
development due to its ability to circumvent the necessity of DC isolation, ex vivo cultivation and re-infusion.
The selection of appropriate antigen of interest remains one of the major challenges for cancer vaccine devel-
opment. The rapid development and large-scale production of mRNA platform has enabled for the development
of both personalized vaccines (mRNA 4157, mRNA 4650 and RO7198457) and tetravalent vaccines (BNT111 and
mRNA-5671). In addition, mRNA vaccines combined with checkpoint modulators and other novel medications
that reverse immunosuppression show promise, however further research is needed to discover which combi-
nations are most successful and the best dosing schedule for each component. Each delivery route (intradermal,
subcutaneous, intra tumoral, intranodal, intranasal, intravenous) has its own set of challenges to overcome, and
these challenges will decide the best delivery method. In other words, while developing a vaccine design, the
underlying motivation should be a reasonable combination of delivery route and format. Exploring various
administration routes and delivery route systems has boosted the development of mRNA vaccines.
Introduction
Vaccinations play a vital role in reducing disease, disability, and
mortality from a variety of infectious diseases [1]. The use of conven-
tional vaccines such as live attenuated vaccines, inactivated pathogens,
subunit vaccines or toxoid vaccines provides durable efcacy against
various infectious diseases [2]. Nucleic acid vaccines mainly, plasmid
DNA(pDNA) and messenger RNA (mRNA), came to existence in 1900s
due to their innate ability to stimulate inoculation with live organism-
based vaccines, notably for cell-mediated immune stimulation [3]. For
several decades later, pDNA-based approaches dominated the eld,
since mRNA-based approach was considered unstable due to inefcient
in-vivo delivery and excessive stimulation of inammatory responses
[4,5]. Eventually in late 2000s, a series of improvement in manufacture,
modication and stabilization of mRNA led to its recognition as a
resourceful platform for developing novel therapy [4,5]. mRNA vaccines
thus hold a lot of promise and confer several advantages over traditional
vaccines.
The recent outbreak of SARS-CoV-2 and coronavirus disease (Covid-
19) has demonstrated an urgent need of rapid vaccine development.
Two mRNA vaccines, BNT162b2 (Pzer-BioNTech) and mRNA-1273
(Moderna TX), have acquired authorization from FDA that are
currently being used to prevent COVID-19 [6]. Both vaccines have good
efcacy as demonstrated in the various phase III trials and real world
studies [7–11]. Knowledge gained from these trials and versatile ther-
apeutic potential of the mRNA can be applied for the development of
vaccine for the infectious diseases and cancer. In this review, we focus
on the therapeutic aspect of mRNA vaccines as a cancer therapy. In
* Corresponding author at: Fosun Pharma USA Inc, 91 Hartwell Avenue, Suite 305, Lexington, MA 02421, USA.
E-mail address: aimin.hui@fosunpharma.com (A.-M. Hui).
Contents lists available at ScienceDirect
Cancer Treatment Reviews
journal homepage: www.elsevier.com/locate/ctrv
https://doi.org/10.1016/j.ctrv.2022.102405
Received 5 February 2022; Received in revised form 25 April 2022; Accepted 29 April 2022
Cancer Treatment Reviews 107 (2022) 102405
2
addition, we would apprise the literature on the various types of cancer
vaccines, the novel platforms available for delivery of the vaccines, the
recent progress in the RNA-based therapies and the evolving role of
mRNA vaccines for various cancer indications, the available clinical and
preclinical studies with the future chapter in treatment of patients.
The available platforms for development of anti-cancer vaccines
Cancer vaccines are a promising new immunotherapeutic strategy
for both prevention and treatment. Vaccines targeting tumor associated
or tumor-specic antigens (TAAs or TSAs) can destroy malignant cells
that overexpress the antigens due to immunologic memory, resulting in
a durable therapeutic response. Compared to other immunotherapies,
cancer vaccines provide a precise, safe, and acceptable treatment.
Currently, 2 prophylactic vaccines have been approved by the U.S. Food
and Drug Administration (FDA) for routine use in clinical practice.
Gardasil-9 is approved for prevention of HPV infection that is the cause
of most HPV cancers. The other one is hepatitis B (HBV) vaccine, for
example HEPLISAV-B, to prevent HBV infection that is known to cause
hepatocellular carcinoma [12,13].
In 2010, PROVENGE (sipuleucel-T), an immune-cell based thera-
peutic cancer vaccine was granted approval for the treatment of
individuals with asymptomatic or mild symptomatic metastatic
castration-resistant prostate cancer (mCRPC) [14]. Besides, therapeutic
vaccines are available for the treatment of early-stage bladder cancer
(TheraCys® and TICE® Bacillus Calmette-Guerin (BCG)) [15] and
melanoma [IMLYGIC® (talimogene laherparepvec/T-VEC)] [16].
Despite signicant attempts to produce cancer vaccines, clinical trans-
lation of cancer vaccines into effective therapeutics has remained dif-
cult for decades due to the wide range of tumor antigens and low
immune response [17], originating the need to develop more potent
vaccine approaches. Furthermore, there is a growing demand for vac-
cine development, large-scale manufacture, and dissemination, partic-
ularly in the case of non-viral diseases such as cancer [18,19].
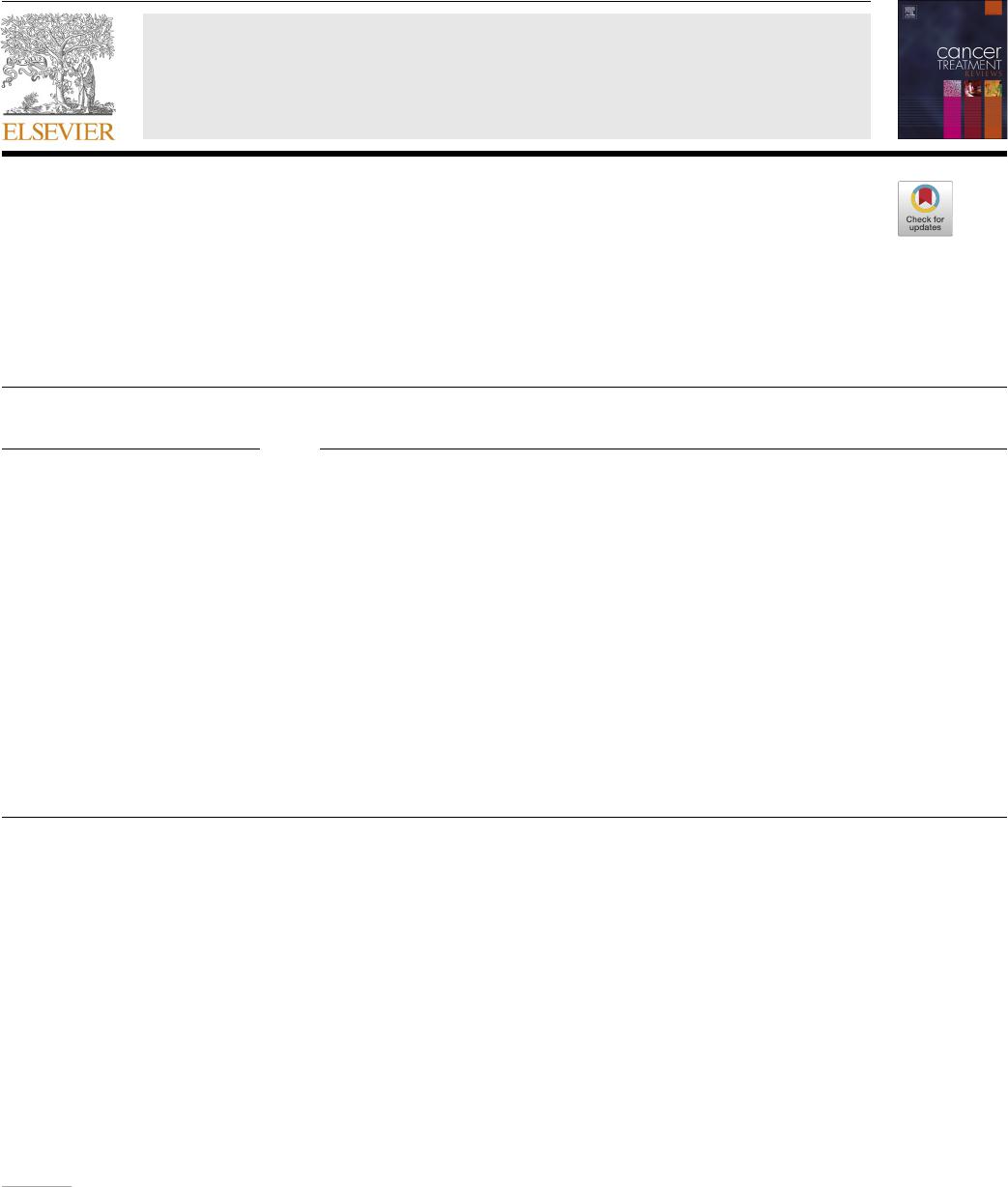
In general, cancer vaccine platforms are classied into tumor cell,
peptide, viral vector, dendritic cell (DC), DNA and RNA types (Fig. 1).
Allogenic or autologous patient-derived tumor cells are used to make
cellular vaccines [20]. This approach is benecial in that target antigens
does not have to be determined in advance [21]. The whole cell cancer
vaccine approach using granulocyte–macrophage colony-stimulating
factor (GM-CSF) has been studied in several types of cancer both in
animals as well as human trials. The phase I and II studies with alloge-
neic GM-CSF–transduced vaccine post-radiation (derived from two
pancreatic tumor lines) demonstrated durable efcacy and prolonged
Fig. 1. The commonly available platforms and mechanisms for cancer vaccine development. (a) Whole cell-based vaccines (an autologous tumor cell vaccine using a
patient’s own cancer cells is injected as vaccine). (b) Viral vector-based vaccines (the genome of viral particles is modied to contain one or more genes encoding for
the antigens of interest). (c) Dendritic cell-based vaccines (the dendritic cells efciently capture the antigens, internalize, and process into peptides that are then
presented in the context of MHC I and II molecules. These complexes are later recognized by the T-cell receptor (TCR) of CD8+ and CD4 + T cells) (d) DNA based
vaccines (DNA plasmids are designed to deliver genes encoding TAs, eliciting or augmenting the adaptive immune response towards TA-bearing tumor cells. It
induces the innate immune response, stimulates several DNA-sensing pathways in the cytosol of transfected cells due to the presence of CpG motifs and the double
stranded structure itself) (e) Peptide-based vaccines (the peptides bind with the restricted MHC molecule expressed in APC. The peptide/MHC complex is then
transported to the cell surface after intracellular processing and later recognized by the TCR on the surface of T cells, leading to activation of T lymphocytes) (f) RNA
based vaccines (conventional non-replicating mRNA consists of 5 structural elements such as cap structures, a 5
′
untranslated region (5
′
-UTR), an open reading frame
encoding antigens of interest, a 3
′
-UTR; and an adenine repeating nucleotide sequence that forms a polyadenine (poly(A) tail. The non-replicating mRNA encodes
antigen of interest, while self-amplifying mRNA encodes antigen of interest and a replication machinery, a self-replicating single-stranded RNA virus).
J. Wei and A.-M. Hui
Cancer Treatment Reviews 107 (2022) 102405
3
survival in patients with pancreatic cancer [22,23].
Peptide vaccines are made up of amino acid sequences that contain
an epitope which can cause an immune response. Due to the difculties
of small peptides to attach directly to major histocompatibility com-
plexes (MHC) I molecules, long peptides (containing of between 25 and
35 amino acids) are frequently favored over short peptides (consisting of
approximately 10 amino acids). Short peptides also fail to activate CD4
helper T cells, which are required for full cytotoxic T lymphocyte acti-
vation (CTLs). These shortcomings can be overcome by using a long-
peptide vaccine, that forces dendritic cells (DCs) to phagocytose the
long-peptide before it is exposed on MHC I and attached to T cells. Long
peptide vaccines also increase the HLA-related compatibility that exist
with short-peptide vaccine. Furthermore, using a long peptide vaccine
permits APCs to be presented via MHC II, which stimulates CD4+
lymphocytes, allowing for a more efcient immune response against
tumor cells. However, because peptides are not self-immunogenic,
administering an adjuvant at the same time is required for producing
maximum efciency [24]. So far, the peptide-based vaccines tested in
laboratory has been able to elicit limited tumor-targeting immune re-
sponses, mostly because of intrinsic changes in cancer cells that reduce
antigenicity and/or changes immunosuppressive alterations in the
tumor microenvironment [25]. Therefore, other approaches are being
developed including its combination with other immunotherapies, tar-
geting antigenic epitopes arising from tumor cells and identifying target
population [25].
Genetically modied viruses are also used for mRNA delivery.
Application of positive strand RNA viruses via translation with host ri-
bosomal machinery. However, challenges with host genome integration
and the likelihood of host rejection, as well as cytotoxicity and immu-
nogenicity, remains the major challenges. The MHC allows cancer cells
to create peptide antigens that are present on their membrane surface. T
cell receptors (TCRs) on cytotoxic T lymphocytes (CTLs) identify these
antigens, resulting in cancer cell lysis. The antiviral immune response
neutralizes viral vectors, limiting the number of vaccines that can be
given [21].
Finally, to boost the adaptive immune system against tumor anti-
gens, DNA cancer vaccines are created from bacterial plasmids (naked
DNA) expressing one or more tumor antigens. The capacity of DNA
vaccines lies in its ability to combine many genes expressing numerous
tumor-antigens to establish a precise and broader adaptive immune
response at the same time. However, these vaccines are poorly immu-
nogenic [24]. To improve the immunological response of DNA vaccines,
researchers have looked into encoding xenogeneic versions of antigens,
fusing antigens with compounds that activate T cells or trigger asso-
ciative recognition, DNA vector priming followed by viral vector
boosting, and immunomodulatory molecules [26]. In contrast, RNA
cancer vaccines are superior to DNA vaccines. While RNA is more sus-
ceptible to RNase breakdown, this can be minimized through chemical
changes and the insertion of modied nucleosides such as pseudo uri-
dine. Furthermore, unlike DNA, which must overcome the second bar-
rier, the nuclear membrane, to reach the nucleus, RNA just needs to
enter the cytoplasm [21]. The encoded proteins are converted into
peptides that are present on MHC I and II to excite CD8+ and CD4+ T
cells, respectively, after RNA translation. The fundamental pharma-
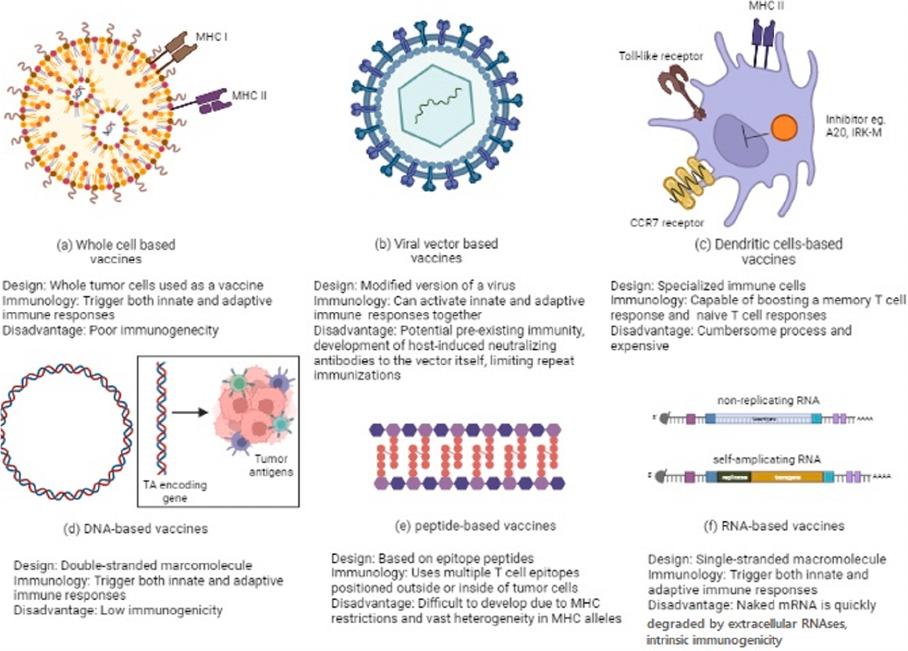
cology of mRNA vaccines is presented in Fig. 2.
Given the importance of DCs in initiating adaptive immunity in vitro
and in vivo through generating CTLs, mRNA-transfected DC vaccine is
an approach gaining interest for cancer vaccine development [24]. DC-
based mRNA cancer vaccines have shown promising effects in various
phases of clinical trials. Boczkowski and colleagues in 1996 rst
demonstrated that electroporation of DCs with mRNA could elicit potent
immune responses against tumor in mice [27]. Since then, several
human trials with electroporation of DCs have been conducted [28,29].
Bulk mRNA isolated from autologous tumors is another method for
pulsing DCs with tumor antigen-loaded mRNA [30,31]. Direct injection
of mRNA can be used instead of DC vaccines since it eliminates the need
for DC isolation, ex vivo cultivation, and re-infusion [32]. Directly
injecting the mRNA into secondary lymphoid tissue aids in delivering
antigen to APCs at the T cell activation site, circumventing the need for
DC movement [33].
Unlocking the potential of mRNA cancer vaccines
The cancer vaccines have the ability to elicit immune response to
Fig. 2. Mechanism of action of mRNA
vaccines. 1. In a cell-free system, mRNA is
in vitro transcribed (IVT) from a DNA
template. 2. IVT mRNA is then transfected
into dendritic cells (DCs) by the process of
(3) endocytosis. 4. Endosomal escape al-
lows entrapped mRNA to be released into
the cytoplasm. 5. The mRNA is translated
into antigenic proteins using the ribosome
translational mechanism. After post-
translational modication, the translated
antigenic protein is ready to act in the cell
where it was produced. 6. The protein gets
secreted by the host cell. 7. Antigen proteins
are digested in the cytoplasm by the pro-
teasome and transferred to the endoplasmic
reticulum, where they are loaded onto MHC
class I molecules (MHC I). 8. MHC I-peptide
epitope complexes with loaded MHC I-
peptide epitopes produced, resulting in in-
duction. 9. Exogenous proteins are taken up
DCs. 10. They are degraded in endosomes
and delivered via the MHC II pathway.
Furthermore, to obtain cognate T-cell help
in antigen-presenting cells, the protein
should be routed through the MHC II
pathway. 11. The generated antigenic pep-
tide epitopes are subsequently loaded onto
MHC II molecules.
J. Wei and A.-M. Hui
Cancer Treatment Reviews 107 (2022) 102405
4
tumor antigens. The selection of a suitable target antigen is pivotal in the
development of a vaccine design. Currently, the majority of vaccinations
are TAAs, which are self-proteins that are improperly expressed by
cancerous cells [21]. Developing vaccines against TAAs is challenging,
as B- and T-cells might be subjected to removal by central and peripheral
tolerance [34]. Besides, along with overexpression on tumor cells, TAAs
might also be expressed in normal healthy cells leading to collateral
damage [21]. In contrast, TSAs, which consists of neoantigens and viral
oncoproteins are expressed only in cancerous cells. The prophylactic
viral oncoproteins work by inducing the production of powerful
neutralizing antibodies that block viral entrance into host cells and
neoplasia caused by viruses [21]. However, these vaccines were inef-
fective in curing cancer as humoral immunity cannot effectively elimi-
nate larger number of virus-infected cancer cells [21]. Neoantigens, like
viral oncoproteins, are specic to tumor cells and are recognized by the
immune system as foreign substances. Lately, neoantigens are being
considered as a potential target in the progress of anti-cancer vaccine
development. Numerous pre-clinical trials and early phase clinical trials
have shown the ability of neoantigen based vaccines to minimize the
potential induction of central and peripheral tolerance as well as the risk
of autoimmunity [35,36].
TAAs with shared expression across cancer types, such as melanoma-
associated antigen (MAGE1) and NY-ESO-1.37, has encouraged studies
to target TAAs that are habitually overexpressed in a certain type of
cancer, along with the prospect of generating a common vaccine per
tumor type [37]. Empirical clinical experience has also suggested that
vaccines targeting specic tumor antigens are ineffective in tackling
tumor heterogeneity, as well as in dealing with the challenges of clonal
evolution and immune evasion by the tumor [38]. As a result, with the
increasing importance of therapeutic cancer vaccines, the rapid devel-
opment and large-scale production using mRNA platform introduces the
potential for the development of both personalized vaccines and off-
shelf cocktail vaccines.
Personalized cancer vaccines (PCV)
The neoantigens remain unique for each individual, with their
numbers varying on the type of cancer. This necessitates for a tailored
approach in which the tumor genome is sequenced and mutations are
detected, neoantigens are predicted using computerized algorithms and
a vaccine is then created and delivered to the patient. Mice vaccinated
with a computationally engineered synthetic mRNA comprising
numerous MHC class II neoepitopes showed 100% tumor rejection in
preclinical studies, demonstrating antigen distribution [39]. The safety
and efcacy of this approach was established in a rst-in-human clinical
study involving 13 patients with metastatic melanoma. Each patient was
given a vaccine that contained 10 neoepitopes specic to their tumor. In
certain patients, antitumor responses were discovered in metastases
removed after immunization, where T-cell inltration and neoepitope-
specic apoptosis of autologous tumor cells were discovered after
vaccination. All patients exhibited CD4+ and CD8+ T-cell responses
[40]. Since then, therapeutic cancer treatment with tailored mRNA
vaccines has received a lot of interest, and several clinical trials are
presently underway, according to the US National Library of Medicine. A
recent study with mRNA-4650, a KRAS personalized vaccine, developed
by Moderna and Merck, in combination with or without pembrolizumab
was conducted to treat patients with pancreatic carcinoma. The lipid
nanoparticles (LNPs) approach for delivery of mRNA-4650 showed well-
tolerated anti-tumoral immune response [41]. Another personalized
vaccine, mRNA-4157, targeting 20 TAAs and useful in treating various
types of tumors, in single or in combination with pembrolizumab
demonstrated acceptable safety prole with cytotoxic T-lymphocyte
(CTL)- and memory T-cell-dependent immune responses [42]. Based on
the ability of mRNA-4157 to elicit clinical response, a phase II trial is
currently undergoing to evaluate the efcacy of the postoperative
adjuvant therapy with mRNA-4157 and pembrolizumab in comparison
with pembrolizumab monotherapy in high-risk recurrent individuals
with complete resection of tumor (NCT03897881). A rst-in-human
phase Ib study of RO7198457, a combination of systemically adminis-
tered RNA-Lipoplex iNeST with the PD-L1 antibody atezolizumab is
presently conducted in patients with locally advanced or metastatic
solid tumors. The preliminary results of this study suggest signicant
level of neoantigen immune-tumor response. A randomized phase II
study of RO7198457 in rst-line for patients with melanoma in combi-
nation with pembrolizumab is currently ongoing, and 2 randomized
clinical trials are planned for the adjuvant treatment of individuals with
non-small cell lung cancer (NSCLC) and colorectal cancer (CRC) [43].
Tetravalent vaccine and combination therapies
A tetravalent RNA-lipoplex cancer vaccine, BNT111, contains 4 types
of naked RNA such as RBL001.1, RBL002.2, RBL003.1, and RBL004.1
encoding 4 melanoma-associated antigens (MAAs), the cancer-testis
antigen NY-ESO-1, the human MAGE- A3, tyrosinase, and putative
tyrosine-protein phosphatase (TPTE), encapsulated in liposomes. The
vaccine upon intravenous administration is taken up by the APCs, and
after being translocated to the cytoplasm, is translated into the 4 tumor-
associated proteins. As a result, CD8+ and CD4+ T-cell responses
against 4 selected antigens are produced [44]. A phase I trial showed
that this vaccine alone and in combination with immune checkpoint
inhibitors (ICIs) induced durable objective responses and exhibited a
favorable safety prole among patients with advanced melanoma [45].
A phase II trial is ongoing to evaluate the vaccine candidate in combi-
nation with the anti-PD-1 antibody cemiplimab for patients with unre-
sectable stage III or stage IV melanoma who are refractory to or relapsed
after anti-PD-1 therapy [46].
mRNA-5671, another tetravalent vaccine, is an LNP-formulated
mRNA-based vaccine that targets 4 of the most frequent KRAS muta-
tions (G12D, G13D, G12C and G12V). APCs take up and translate
mRNA-5671 after immunization. Following translation, the MHCs dis-
plays the epitopes on the surface of APCs, resulting in the development
of both cytotoxic T-lymphocyte- and memory T-cell-dependent immune
responses directed at tumor cells with KRAS mutations [47]. CD8 T cell
responses to KRAS antigens were considerably improved in preclinical
investigations after immunization with mRNA encoding KRAS muta-
tions [48]. Patients with advanced or metastatic NSCLC, colorectal
cancer, or pancreatic adenocarcinoma and KRAS mutations are being
enrolled in a phase I research using mRNA-5671 with or without pem-
brolizumab (NCT03948763).
Due to heterogenous and ever evolving nature of cancer mechanisms,
the clinical benet of monotherapy regimen in patients with advanced
cancer is not adequate. Tumor-specic T lymphocytes produced by
vaccines do not operate efciently against the tumor due to their lack of
motility and/or gradual depletion. As a result, combining procedure that
prevent immune escape pathways is critical [49]. For instance, a phase II
clinical trial in chemotherapy treated patients with metastatic
castration-resistant prostate cancer (mCRPC) showed similar and dura-
ble tumor immune responses on addition of DC vaccines [50]. Mono-
clonal antibodies (mAbs) targeting CTLA-4 and the PD-1/PD-L1
expression have revolutionized the treatment paradigm for several types
of cancers, including renal cancer, melanoma, bladder cancer, lung
cancer and Hodgkin’s lymphoma [51]. CureVac GmbH systemic mRNA
immunotherapy and local irradiation therapy can eradicate established
macroscopic E.G7-OVA and LLC cancers in a synergistic manner.
Moreover, this combination boosted CD4
+
, NKT and CD8
+
cell inltra-
tion in tumor infected mouse [52]. CV9202, vaccine encoding 6 NSCLC-
associated antigens (NY-ESO-1, MUC-1, MAGE-C2, MAGE-C1, 5T4 and
survivin) have been proven to induce targeted immune responses. The
combination of this vaccine with radiotherapy in a phase Ib clinical trial
in 26 stage IV NSCLC patients revealed elevated CV9202 antigen-
specic immune responses in 84% of patients, with 80% increased
antigen-specic antibody levels, 40% patients with functional T cells
J. Wei and A.-M. Hui

Cancer Treatment Reviews 107 (2022) 102405
5
and about 52% of patients had multiple antigen specicities [53]. In
another study, researchers used an mRNA vaccine expressing the TAA
MUC1 in combination with an anti-CTLA-4 monoclonal antibody to
boost the vaccine’s immune response against triple-negative breast
cancer (TNBC) by improving T cell activity [54].
Recent advancement of mRNA vaccines in various types of
cancer
Preclinical and clinical evidence have shown that using mRNA for
prophylaxis and therapy can help prevent infectious disease and treat
cancers, and that mRNA vaccines are safe and well tolerated in both
animal models and humans. Further enhancements might also boost
antigen-specic immune responses as well as B and T cells immune re-
sponses [55]. As of 21st December 2021, 23 RNA vaccines are currently
under phase I/II/III clinical trials, while 24 vaccines are at pre-clinical
stage.
Breast cancer
Breast cancer remains a cause of mortality for women globally [56].
More often, 81% women suffer from invasive breast cancer, which
comprises of at least 21 distinct histological subtypes and 4 molecular
subgroups (luminal A, luminal B, triple-negative and HER2-enriched)
that differ in risk factors, presentation, response to treatment, and out-
comes [57]. Invasive breast cancer can spread to adjacent lymph nodes
or other organs over time. It is because of widespread metastasis that a
woman dies from breast cancer [58]. Using modern methodologies for
mRNA sequencing, such as The Cancer Genome Atlas (TCGA) data, it has
been established that increased expression of T- and B-cell predicts
higher overall survival (OS) in a variety of tumor types, including breast
cancer [59]. The current treatment approach for breast cancer includes
radiation therapy, surgery, chemotherapy, as well as hormonal and
targeted therapies. Lately, the development of medications that can
prevent breast cancer from developing in the rst place, as well as their
recurrence, has gathered attention. The overexpression of high-afnity
transmembrane receptors such as HER3, HER2, c-MET, EGFR, and the
transmembrane protein epithelial mucin-1 (MUC-1) are the key onco-
genic drivers for breast cancer [60]. Treatment of breast cancer, espe-
cially, TNBC is gaining importance, since lack of therapeutic targets
makes such type of cancer unresponsive to typical endocrine therapies
and HER2-targeted therapy. In such a case, cancer vaccines which aid in
activation and amplication of TAA-specic immunity combined with a
sustained memory T cell immune response may be an effective therapy
for preventing breast cancer recurrence in patients [61]. Previous
vaccination strategies in adjuvant settings, against HER2+ self-antigens
have shown substantial efcacy in patients with breast cancer [62–64].
However, such an approach is usually weak as immune response as T-
lymphocytes have afnity to HER2+ and thus are subject to central
tolerance [65]. An ongoing phase I/II trial is being conducted in patients
with TNBC and who completed standard of care chemotherapy, where
patients are allocated to receive either 8 vaccination cycles of mRNA
WAREHOUSE vaccine (containing pre-formulated, shared tumor anti-
gens, non-mutated) or mRNA MUTANOME vaccine (containing indi-
vidual mutations). The preliminary data of this trial showed mRNA
WAREHOUSE is feasible approach for treatment of TNBC [66]. Another
phase I trial from the Schmidt and colleagues was conducted with the
addition of a third arm where patients were injected with IVAC_M_uID
[Individualized NeoAntigen Specic Immunotherapy (iNeST)] which
encodes 20 cancer mutations neoepitopes derived from NGS. The initial
results reported promising results of iNeST IVAC_M_uID in inducing
strong polyepitope T-cell responses in patients with TNBC in the post-
(neo)adjuvant phase or post-surgery. All the patients reported CD4+
and/or CD8+ T-cell responses against 1 to 10 of the vaccine neoepitopes
[67]. Theoretically, this treatment regimen will lead to a transition from
an individualized therapy targeting a single biomarker (e.g., HER2) to a
fully specialized treatment targeting specic mutations in each patient.
The ongoing trials related to mRNA vaccine in breast cancer is listed in
Table 1.
Non-small cell lung cancer
Lung cancer remains a major cause of cancer worldwide after breast
cancer. Despite recent therapeutic advancements, the overall 5-year
survival rate for LC is still less than 20%. Because most cancers exhibit
mutational variability, conventional cancer treatment techniques, such
as surgery and chemotherapy, are far from optimum, especially for
advanced stage malignancies. Currently, the information related to
mRNA-based approach in treatment for NSCLC is limited. CV9201 is a
cancer immunotherapy based on RNActive® that encodes 5 NSCLC an-
tigens: melanoma antigen family C1/C2, NY esophageal squamous cell
carcinoma-1, trophoblast glycoprotein and survivin. About 46 patients
with locally advanced (n = 7) or metastatic (n = 39) NSCLC received 5
intradermal CV9201 injections (400–1600 g of mRNA) in a phase I/IIa
dose-escalation experiment. After initial dose administration, the me-
dian progression-free survival and OS were 5.0 months (95 percent CI
1.8–6.3) and 10.8 months (8.1–16.7), respectively. In addition, 60% of
patients reported an increased frequency of >2 fold followed by acti-
vation of IgD+ CD38hi B cells. This showed that CV9201 was well
tolerated, and immunological responses could be observed following
therapy, indicating that further clinical research is warranted [68]. The
ongoing trials related to mRNA vaccine in breast cancer is listed in
Table 2.
Prostate cancer
The standard treatment for prostate cancer includes androgen
deprivation and chemotherapy. However, patients become resistant
after prolonged treatment with these agents. Relapse or progression of
disease occur even after complete androgen blockage and when plasma
concentrations of testosterone are reduced to <50 ng/dL by castration or
gonadotropin-releasing hormone analogs, and the effects of the
remaining androgens are suppressed by androgen receptor antagonists
[69]. With the advent of Sipuleucel-T, a dendritic-cell based vaccine, for
treatment of advanced stages of prostate cancer, immunotherapy for
prostate cancer has come into limelight. However, besides sipuleucel-T,
there have been disappointing results in prostate cancer. In patients with
mCRPC, large phase III studies of the CTLA-4 inhibitor, ipilimumab did
not show signicant benet in OS compared to placebo before or after
chemotherapy treatment. In addition, nivolumab, a single-agent PD-1
antibody, was found to have little effect in men with mCRPC. However,
administering both CTLA-4 and PD-1 inhibitors combination has resul-
ted in some PSA and objective responses, showing that a minority of
patients may benet. Pembrolizumab was given to mCRPC patients who
were advancing on enzalutamide in a recent study, and a signicant
number of men had remarkable PSA and objective responses. PSA and
objective responses appeared to be more common in another small trial
Table 1
Clinical trials for breast cancer.
Conditions NCT number Study
design
Interventions Status
Triple
negative
Breast
Cancer
NCT02316457 Phase I IVAC_W_bre1_uID/
IVAC_M_uID
Active, not
recruiting
Breast
Cancer
NCT00003432 Phase
I/II
carcinoembryonic
antigen RNA-pulsed
DC cancer vaccine
Terminated
Breast
Cancer
NCT03788083 Phase I Trimix mRNA Recruiting
Breast
Cancer
NCT03739931 Phase I mRNA-2752/
Durvalumab
Recruiting
J. Wei and A.-M. Hui
