
REVIEW
The Development of mRNA Vaccines for Infectious
Diseases: Recent Updates
Nitika
1,2
Jiao Wei
1,2
Ai-Min Hui
1,2
1
Fosun Pharma USA Inc., Boston, MA,
USA;
2
Shanghai Fosun Pharmaceutical
Industrial Development, Co., Ltd.,
Shanghai, People’s Republic of China
Abstract: mRNA-based technologies have been of interest for the past few years to be used
for therapeutics. Several mRNA vaccines for various diseases have been in preclinical and
clinical stages. With the outbreak of the COVID-19 pandemic, the emergence of mRNA
vaccines has transformed modern science. Recently, two major mRNA vaccines have been
developed and approved by global health authorities for administration on the general
population for protection against SARS-CoV-2. They have been proven to be successful in
conferring protection against the ongoing SARS-CoV-2 and its emerging variants. This will
draw attention to various mRNA vaccines against infectious diseases that are in the early
stages of clinical trials. mRNA vaccines offer several advantages ranging from rapid design,
generation, manufacturing, and administration and have strong potential to be used against
various diseases in the future. Here, we summarize the mRNA-based vaccines in develop
-
ment against various infectious diseases.
Keywords: vaccines, mRNA, infectious diseases, lipid nanoparticles, immune response
Introduction
Vaccines are important for protection against infectious agents and confer protec-
tion against various diseases to humans
1
. Vaccines have been prominent in eradi-
cating various deadly diseases such as smallpox, polio, measles, mumps, rubella,
and other such infections.
2–4
Conventionally, vaccines were developed using che-
mical or heat treatment and live attenuated pathogens in cell lines or animals. While
live attenuated vaccines (LAV) have been successful in the eradication of numerous
deadly diseases, their safety and efcacy remain questionable in some diseases.
2
Some disadvantages of live attenuated vaccines include their failure if there are
mutations in the surface antigens of the pathogens, causing the disease in immune-
compromised individuals and chances of reversion to a virulent form due to reverse
mutations in the pathogen.
5,6
These vaccines are less efcient in mounting CD8
+
immune response, which is important for protection against bacteria and viruses.
7
Recently, nucleic acid therapeutics have emerged as an alternative to conven-
tional vaccines. One of the nucleic acid vaccine approaches involves the develop-
ment of messenger ribonucleic acid (mRNA) vaccines.
8
The rst mRNA vaccine
was developed in rats to reverse diabetes insipidus using intrahypothalamic injec
-
tion of vasopressin mRNA.
9
mRNA vaccines are thought to be benecial in
comparison to the conventional vaccines due to the non-infectious and non-
integrative nature of mRNA in the human body.
10
Additionally, mRNA half-life
can be modulated in a cell by adding modications onto the mRNA molecule.
10
Correspondence: Ai-Min Hui
Shanghai Fosun Pharmaceutical Industrial
Development, Co., Ltd., 1289 Yishan
Road, Shanghai, 200233, People’s Republic
of China
Tel +86-21-33987000
Fax +86-21-33987020
Email aimin.hui@fosunpharma.com
Infection and Drug Resistance 2021:14 5271–5285
5271
© 2021 Nitika et al. This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.
php and incorporate the Creative Commons Attribution – Non Commercial (unported, v3.0) License (http://creativecommons.org/licenses/by-nc/3.0/). By accessing the
work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For
permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms (https://www.dovepress.com/terms.php).
Infection and Drug Resistance Dovepress
open access to scientific and medical research
Open Access Full Text Article
Received: 4 October 2021
Accepted: 25 November 2021
Published: 9 December 2021
Infection and Drug Resistance downloaded from https://www.dovepress.com/ by 108.7.72.234 on 09-Dec-2021
For personal use only.
Powered by TCPDF (www.tcpdf.org)

Nucleic acid-based vaccines mimic infection with live
pathogens and stimulate follicular T helper and germinal
B cell immune response.
11
Here we review different types of mRNA vaccines,
delivery, and immune responses generated by mRNA vac
-
cines and various mRNA-based vaccines against infectious
diseases in clinical trials.
Types of mRNA Vaccines
Currently, there are two forms of mRNA vaccines: non
replicating mRNA vaccines (modied and unmodied
mRNA (NRM)) and self-amplifying mRNA (SAM) vac
-
cines, which are derived from positive-strand RNA
virus.
12–17
mRNA vaccines are produced by articially
synthesizing mRNA via a cell-free in vitro enzymatic
transcription reaction. This in vitro reaction consists of
linearized plasmid DNA, RNA polymerase and nucleoside
triphosphates. NRM vaccines consist of an open reading
frame (ORF) for a target antigen, untranslated regions
(UTRs) at both ends and a terminal poly A tail.
18
Once
inside the body, they drive antigen expression. Naked
unmodied mRNA is prone to be degraded as soon as it
is delivered inside the body. However, it was discovered
that incorporation of naturally occurring chemically mod
-
ied nucleosides such as pseudouridine and 1-methylpseu-
souridine results in the efcient translation of RNA.
19,20
Another mRNA vaccine platform is based on alphavirus,
a positive-strand virus.
21
In these vaccines, the structural
proteins are replaced with the gene of interest. These self-
amplifying mRNA can direct self-replication RNA-
dependent RNA polymerase complex and generate multiple
copies of the antigen-encoding mRNA.
22
They express high
levels of the heterologous gene when introduced into the
cytoplasm of host cells. They mimic the production of anti
-
gens in vivo by viral pathogens and trigger both humoral and
cellular immune responses.
22
SAMs produce higher levels of
antigens as compared to NRM vaccines. Both NRM and
SAM vaccines do not integrate into the host genome.
mRNA vaccines are much safer than other vaccine platforms
and hold promise to be effective against infectious
pathogens.
13,15,20,23,24
Delivery of mRNA Vaccines
The delivery of mRNA vaccines is crucial due to their
unstable nature. Intravenous administration of unmodied
mRNA leads to its digestion by ribonucleases and stimu
-
lates innate immune response.
25,26
The immune system
cells including antigen-presenting cells (APCs), B cells
and T cells should be stimulated by mRNA delivery.
27–29
Delivery of mRNA molecules is challenging as mRNA
molecule is 4 times larger than other molecules. In addi
-
tion, mRNA is negatively charged and repulsed by the cell
membrane.
30
This limitation can be overcome by using
modied RNA and better delivery systems. Various
mRNA platforms have been designed including protamine,
dendrimers, polyethyleneimine and lipid nanoparticles.
31
Lipid nanoparticles (LNP) loaded with nucleoside modi-
ed NRM is used often for administering these vaccines
generally in clinical trials.
31
LNPs are composed of phos-
pholipids, cholesterol, ionizable lipids and lipid-anchored
polyethylene glycol (PEG).
32
LNPs help cellular uptake,
aid endosomal uptake, and enhance cytoplasmic
delivery.
33
LNPs also protect mRNA from being recog-
nized by endosomes and toll-like receptors (TLRs) and
prevent overactivation of the innate immune system.
34
Some mRNA vaccines have adjuvant properties, which
can be benecial.
35
mRNAs can be modied to enhance
immune activation without blocking mRNA expression.
This includes modication of a nucleoside with a TLR-4
agonist monophosphoryl lipid A (MPLA) which aids in
the induction of T cell activation.
36,37
Another strategy
employed is using a short double-stranded region in
poly-A tail or 3ʹUTR of mRNA. The addition of a short
poly U or poly-A tail induces IFN-B and IL-6 and
enhances dendritic cell activation and migration. The ds
poly U or poly-A tail is recognized by TLR3 and RIG-
I.
10,38
Recently, two SARS-CoV-2 RNA lipid nanoparticle
vaccines based on nucleoside modied mRNA (modRNA)
were designed.
39,40
BNT162b1 is a modRNA with blunted
immune sensor activating capacity and enhanced RBD
expression.
41
BNT162b2 is a modRNA vaccine that
expresses P2 mutant, prefusion spike glycoprotein (P2 S)
(version 9).
42,43
Immune Response to mRNA
Vaccines
The mechanism of immune response to mRNA vaccine is
still being studied. In humans, there are two kinds of RNA
sensors the RIG-I-like receptor family and TLRs.
38
There
are four TLRs: TLR3, TLR7, TLR8 and TLR9 which are
present in dendritic cells, macrophages, and monocytes.
TLR3 recognizes double-stranded RNA (dsRNA) and sin
-
gle-stranded RNA (ssRNA). TLR7 binds to both dsRNA
and ssRNA and TLR8 recognizes ssRNA only.
44,45
The
RIG-I family includes RIG-I, MDA-5 and LGP2. RIG-I
https://doi.org/10.2147/IDR.S341694
DovePress
Infection and Drug Resistance 2021:14
5272
Nitika et al Dovepress
Powered by TCPDF (www.tcpdf.org)
Infection and Drug Resistance downloaded from https://www.dovepress.com/ by 108.7.72.234 on 09-Dec-2021
For personal use only.
Powered by TCPDF (www.tcpdf.org)

recognizes ssRNA and dsRNA and stimulates interferon
production.
46,47
MDA5 is a cytosolic RNA sensor that
detects long ds RNA generated during viral RNA replica
-
tion. This recognition of ds RNA activates IRF-3 and NF-
KB and increased the production of IFN-I.
48,49
The
induction of interferons (IFNs) by mRNA vaccines
depends on the in vitro transcribed mRNA, administration
route and the delivery vehicle.
50
Post-mRNA vaccination,
pattern recognition receptors (PRRs) are activated, and
there is enhanced type I IFN production.
51
The IFN pro-
duction can be positive via activation of the immune
response or negative by blocking mRNA translation.
52
Vaccine-Induced Immunity
A cellular immune response such as cytotoxic T cells (CTLs)
directly targets the viral-infected cells in contrast to neutra
-
lizing the virus, which is achieved by humoral immunity.
53,54
The level of antibody generation depends on the size of
LNPs which are transport to the draining lymph nodes,
thus activating B cells. B cells can uptake the LNPs with
mRNA and facilitate the production of protein from the
mRNA.
55
In addition, B cells interact with the B cell recep-
tors expressing the foreign antigens. B cells located in the
draining lymph nodes (with the LNP-mRNA) secrete speci
-
c low-afnity antibodies and some may enter a germinal
center (GC).
56
B cells that enter GC undergo somatic hyper-
mutation (SHM) and afnity maturation. The afnity
matured B cells later become plasmablasts and secrete high-
afnity antibodies. However, they can also re-enter GCs that
undergo SHM and become memory B cells
57
(Figure 1).
For efcient CTL induction, antigen should enter the
route of antigen processing. The pathogenic antigens are
then delivered to the cytosol for proteasomal processing.
Later, the peptides generated from the proteasomal path
-
way are delivered to the endoplasmic reticulum (ER) by
the transporter associated with antigen processing (TAP).
58
In the ER, these peptides bind to the major histocompat-
ibility complex class I (MHC). The MHC I-antigen pep-
tide complex is then recognized by the CD8
T lymphocytes at the cell surface.
59
mRNA-based vac-
cines are particularly suitable for the generation of potent
CTL responses because they can express the antigen in the
cytosol of the antigen-presenting cells.
60,61
Antigen Selection
While designing mRNA-based vaccines, various factors
such as antigen selection, vaccine platforms, vaccine
routes and regimen must be taken into consideration.
Antigen selection is a crucial step for vaccine design.
62
In the case of SARS-CoV-2, the structural proteins are
S protein, N protein, matrix protein (M) and envelope
protein (E). N protein is responsible for coating the large
positive-strand RNA genome encased in a lipid envelope
derived from the host cell membrane. S, M and E proteins
are inserted into this membrane.
63
It has been found that in
SARS-CoV-2 only the antibodies against the S protein can
neutralize the virus and prevent infection. Therefore, most
of the vaccines for SARS-CoV-2 are directed against the
S1 domain of the receptor-binding domain.
64
RNA Vaccines in Infectious Diseases
SARS-CoV-2
COVID-19 is caused by SARS-CoV-2 (severe acute
respiratory syndrome–coronavirus 2) that causes respira
-
tory illness, which in severe cases can lead to death.
65
Bats
are the main host of coronavirus and when transmitted to
humans
66
; it is primarily spread via direct transmission.
Coronavirus is composed of spike protein (S), envelope
protein (E), membrane protein (M), nucleocapsid protein
(N) and hemagglutinin esterase dimer protein (H) which
are bound to negative sense RNA.
67
COVID-19 is an
infectious disease and emerged as a global pandemic in
December 2019. Therefore, there is an urgent need to
develop a vaccine against the SARS-CoV-2 virus.
Various COVID-19 vaccines have been in development
since the beginning of the pandemic.
68
BNT162b2 is
a lipid nanoparticle-formulated nucleoside-modied RNA
vaccine. It encodes a prefusion stabilized, membrane-
anchored SARS-CoV-2 full-length spike protein. This
vaccine was approved by the FDA in December for
administration on the public after it showed 95% efcacy
in Phase 3 clinical trials.
69,70
A few weeks later,
mRNA1273, which is also a lipid nanoparticle-
encapsulated mRNA-based vaccine encoding the prefusion
stabilized full-length spike, was approved by the FDA
after its efcacy was 94.1% in phase 3 clinical trials
(Table 1).
71,72
Currently, CV2nCoV containing sequence-
optimized unmodied mRNA coding for a stabilized form
of spike protein encapsulated in lipid nanoparticles (LNP)
remains in preclinical development. CV2nCoV elicits high
titers of cross-neutralizing antibodies against the B.1.1.7,
B.1.1.298 and B.1.351 variants in rat.
73
CV2nCoV is opti-
mized to have enhanced translation, immunogenicity, and
1.8-fold higher protein expression than the previous can
-
didate vaccine CVnCoV.
73,74
CVnCoV showed 47%
Infection and Drug Resistance 2021:14
https://doi.org/10.2147/IDR.S341694
DovePress
5273
Dovepress Nitika et al
Powered by TCPDF (www.tcpdf.org)
Infection and Drug Resistance downloaded from https://www.dovepress.com/ by 108.7.72.234 on 09-Dec-2021
For personal use only.
Powered by TCPDF (www.tcpdf.org)
efcacy against SARS-CoV-2 of any severity in Phase II/
III clinical trials and failed to meet prespecied statistical
success criteria
75
(NCT04652102). It has been now with-
drawn from the regulatory review due to a potential over-
lap with approval timelines for CV2nCoV (CureVac).
ARCoV is another such lipid nanoparticle-
encapsulated mRNA (mRNA-LNP) encoding the recep
-
tor-binding domain (RBD) of SARS-CoV-2 is in Phase
III clinical trials (NCT04847102). ARCoV elicited high
SARS-CoV-2 specic IgG antibodies and strong virus
neutralization titers in mice and cynomolgus
monkeys.
76
mRNA vaccines can be generally designed faster than
any other platform. This is evident from the SARS-CoV-2
nucleoside modied mRNA-LNP vaccine for human use in
42 days post obtaining the SARS-CoV-2 sequence by Pzer-
BioNTech and Moderna.
77,78
mRNA-based vaccines may
prove to be better than other platforms and efcacious against
the mutant virus strains as the mRNA can be designed for
a mix of multiple sequences for a broad coverage during
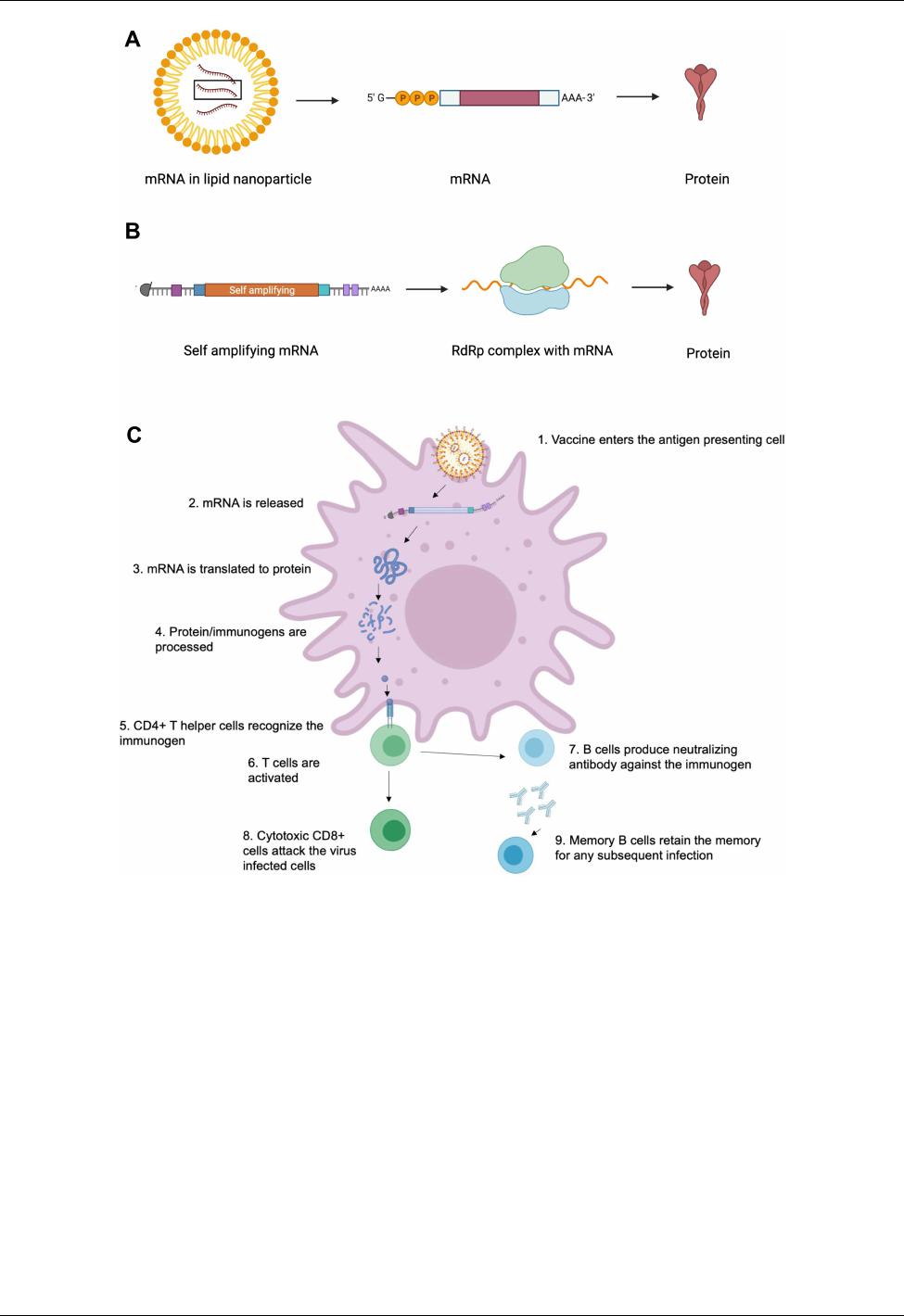
Figure 1 The mechanisms of mRNA vaccine. (A) mRNA vaccine in lipid nanoparticle when released in the body is translated to protein which then elicits immune response.
(B) Self amplifying mRNA is released into the body and then complexed with RdRp which is then translated into protein and elicits immune response. (C) mRNA vaccine
elicits immune response as the protein is expressed and recognized by CD4+ T helper cells; this activates CD8+ T and B cells. B cells produce neutralizing antibodies and
memory B cells retain the memory for any subsequent infection. CD8+ T cells attack virus-infected cells. Created with BioRender.com.
https://doi.org/10.2147/IDR.S341694
DovePress
Infection and Drug Resistance 2021:14
5274
Nitika et al Dovepress
Powered by TCPDF (www.tcpdf.org)
Infection and Drug Resistance downloaded from https://www.dovepress.com/ by 108.7.72.234 on 09-Dec-2021
For personal use only.
Powered by TCPDF (www.tcpdf.org)

Table 1 mRNA Vaccines Against Infectious Diseases
Disease Vaccine/Funding
Source
Dose
Regimen
Phase Outcome/Efcacy Platform References
SARS-CoV-2 BNT162b2, Pzer-
BioNTech
Two-dose
regimen, 21
days apart,
delivered in
the deltoid
muscle
Approved 95% (95% CI, 90.3 to 97.6) LNP- encapsulated full-
length spike mRNA
[69]
SARS-CoV-2 mRNA-1273,
Moderna
Two-dose
regimen, 21
days apart,
delivered in
the deltoid
muscle
Approved 94.1% (95% CI, 89.3 to
96.8%)
LNP- encapsulated full-
length spike mRNA
[88]
SARS-CoV-2 CV2nCoV/CVnCoV,
CureVac AG
Two-dose
regimen, 21
days apart,
delivered
intramuscularly
Preclinical N/A LNP- encapsulated
sequence optimized spike
mRNA
[140]
SARS-CoV-2 ARCoV, Walvax
Biotechnology, PLA
Two-dose
regimen, 28
days apart,
delivered
intramuscularly
Phase III N/A LNP- encapsulated full-
length RBD mRNA
NCT04847102
Rabies CV7201, CureVac
AG
Three-dose
regimen,
delivered
intradermally or
intramuscularly
Phase I GMT of 0·73 IU/mL (95%
CI 0·36–1·46)
LNP- encapsulated full-
length RABV-G mRNA
[96]
Rabies CV7202, CureVac
AG
Three-dose
regimen,
delivered
intramuscularly
Phase I GMTs of 4186 U/mL
(13,253–88,185) and 20,707
U/mL (5592–76,678) for 1
and 2 μg groups
LNP- encapsulated full-
length RABV-G mRNA
NCT03713086
[95]
Inuenza mRNA-H10N8,
mRNA-H7N9,
Moderna
Two-dose
regimen,
delivered
intramuscularly
Phase I For H10N8 HAI GMT was
13.9, for H7N9 HAI GMT
was 13.6
LNP- encapsulated full-
length HA glycoprotein
mRNA from the H10N8
and H7N9 strain
[104]
Respiratory
syncytial virus
(RSV)
mRNA-1345,
Moderna
Three-dose
regimen,
delivered
intramuscularly
Phase I GMF of 20.5 for RSV-A
and GMF of 11.7 for RSV-
B for young adults, GMF of
14.2 for RSV-A and GMF
of 10.1 for RSV-B for older
adults
LNP- encapsulated
prefusion F protein
ModernaTx
NCT04528719
Human
metapneumovirus
(HMPV) and
parainuenza virus
type 3 (PIV3)
mRNA-1653,
Moderna
Three dose
regimen,
delivered
intramuscularly
Phase Ib GMT of 6.04 for HMPV-A,
6.33 for HMPV-B and 3.24
for PIV3
LNP- encapsulated
F protein coding mRNA
from hMPV and PIV3
ModernaTx
NCT04144348
NCT03392389
(Continued)
Infection and Drug Resistance 2021:14
https://doi.org/10.2147/IDR.S341694
DovePress
5275
Dovepress Nitika et al
Powered by TCPDF (www.tcpdf.org)
Infection and Drug Resistance downloaded from https://www.dovepress.com/ by 108.7.72.234 on 09-Dec-2021
For personal use only.
Powered by TCPDF (www.tcpdf.org)
